Regularly monitoring EC and pH in your growing medium can prevent almost all nutrient-related issues in most plants. These parameters give a clear indication of nutrient availability in both soil and soilless environments, and allow you to take corrective measures before such problems affect plant growth and quality.
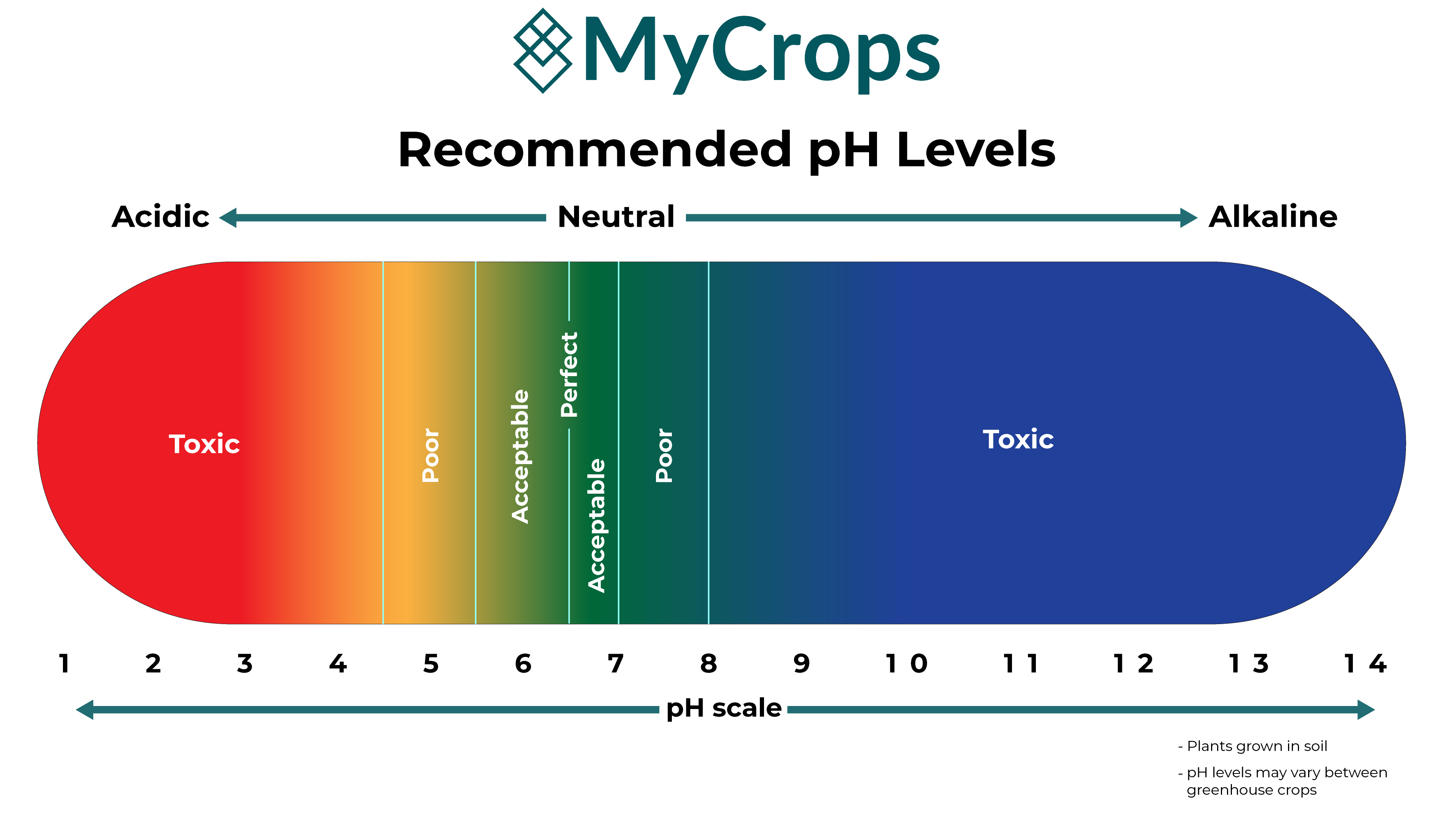
pH is a quantitative measure of the acidity or alkalinity of liquid solutions. The term translates the values of the hydrogen ion concentration into numbers ranging between 0 (acidic) and 14 (alkaline). pH affects myriad processes and properties of soil which determine a plant’s wellbeing. It is adjusted by the concentration of nutrients in the watering mix.
Electrical conductivity (EC) is a measure of the total amount of salts, including fertilizer salts, in the growing medium. Most commonly, conductivity is expressed in MilliSiemens per centimeter (mS/cm). In many cases, conductivity is linked directly to the total dissolved solids (TDS), as TDS level estimations are based on the conductivity of the water. Many growers check these values using a dedicated TDS meter, which measures EC levels and then converts them into a TDS measurement.
Ppm (parts per million) is the most commonly used scale for measuring TDS. Since most salts in fertilizers are macronutrients, EC can be used to indicate the presence of macronutrients in the growing medium. EC is an important indicator of soil health, as it affects crop yields, crop suitability, and plant nutrient availability.
As discussed, pH measures only hydrogen ions, and EC measures the concentration of positively and negatively charged ions in the solution. Any hydrogen present in a solution will impact the pH level and will influence conductivity levels. However, hydrogen ions make up only a small part of the ion concentration measured by a conductivity meter. Their relative relevance depends on the presence of hydrogen in comparison to other concentrations of non-pH determining ions.
The primary nutrients provided in a controlled growing environment are Nitrogen (N), Phosphorus (P) and Potassium (K). So in most cases, EC measurement indicates the relative concentration of these nutrients. Applying this concept helps to estimate the accuracy of the fertilizer concentration applied in a solution.
pH levels strongly affect nutrient and water imbalance. Each nutrient is available to the plant at a different acidity level. The ideal pH will maximize desired nutrient uptake, a value that will change throughout the different grow stages of the plants.
Hard water (120ppm or more) refers to high concentrations of alkalinity (Calcium (Ca), Magnesium (Mg) or Sodium (Na)). When using hard water for irrigation, it’s important to notice the strong effect it has on the EC level of the fertilizer solution. On the other hand, soft water (60ppm less than) contains a small number of dissolved salts and will have marginal effect on the EC values.

Using suitable meters makes it easier to keep track of your soil’s pH and EC parameters. pH and EC meters can be used to check irrigation water quality, calibrate fertilizer, and measure nutrient availability for your plants.
Here are a few important things to note when choosing a pH/EC meter:
2026 MyCrops Technologies Ltd. - All Rights Reserved.